1. Introduction
2. Model and scheme
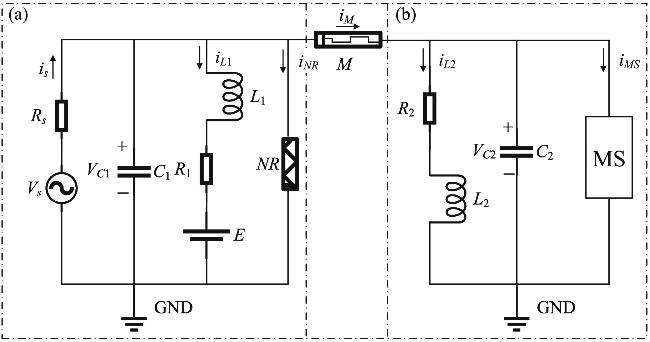
Figure 1. Equivalent circuit diagram of (a) neuron and (b) skeletal muscle cell connected by memristive synapses. |
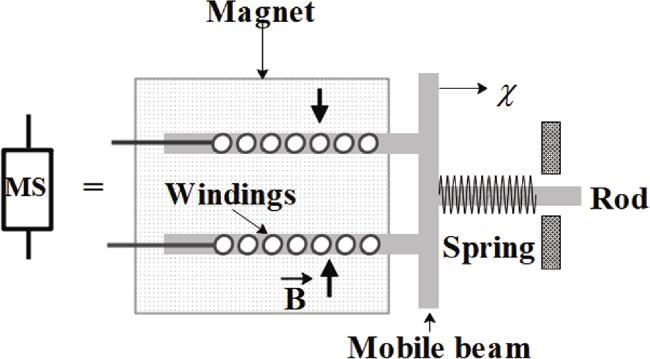
Figure 2. Schematic diagram of the internal structure of an electromagnetically driven robotic arm. |
3. Numerical results and discussion
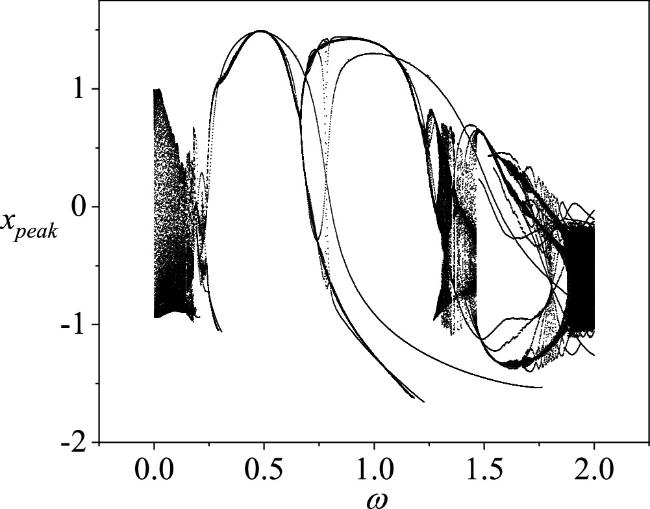
Figure 3. A bifurcation diagram of the membrane potential of a single neuron as the angular frequency ω varies, with the amplitude A set to 0.4. The parameters are chosen as a = 0.7, b = 0.3, c = 0.4, ξ = 0.175, I0 = 0.1. The initial conditions for the single neuron circuit are (0.2, 0.1). |
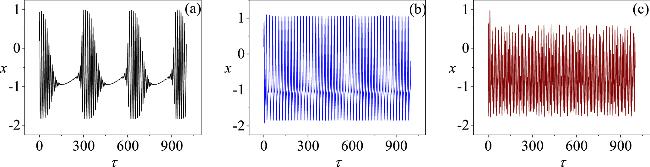
Figure 4. The patterns exhibited by an isolated neuron with different selections of angular frequency ω: (a) clustered firing at ω = 0.02; (b) spike firing at ω = 0.3; (c) chaotic firing at ω=1.3. Other parameters are held constant at a = 0.7, b = 0.3, c = 0.4, ξ = 0.175, I0 = 0.1, A = 0.4, with the initial conditions of the neuron circuit set to (0.2, 0.1). |
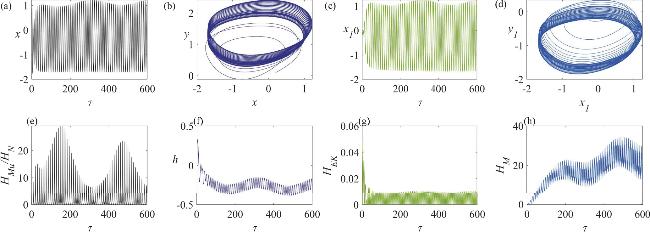
Figure 5. The dynamic behavior of a neuron circuit and a skeletal muscle cell circuit in a state of clustered firing, coupled via memristive synapses: (a) the variation in the neuron’s membrane potential; (b) the dynamics of the neuron circuit’s attractor; (c) the variation in the skeletal muscle cell’s membrane potential; (d) the dynamics of the skeletal muscle cell’s attractor; (e) the temporal evolution of the Hamiltonian energy ratio between the skeletal muscle cell and the neuron; (f) the temporal evolution of the displacement of the active beam; (g) the temporal evolution of the kinetic energy of the active beam; (h) the temporal evolution of the field energy in the memristor-coupled channel. In this analysis, the angular frequency is set to ω = 0.02. |
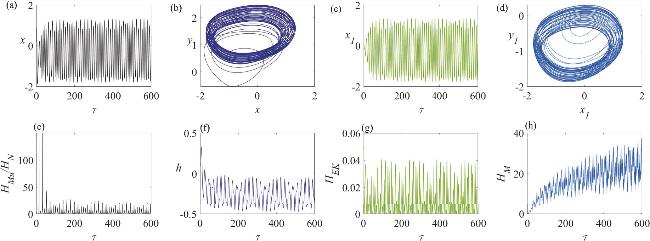
Figure 6. The dynamic characteristics of a neuron circuit and a skeletal muscle cell circuit in a spiking firing state, under the influence of memristive synapse coupling: (a) the variation in the neuron’s membrane potential; (b) the dynamics of the neuron circuit’s attractor; (c) the variation in the skeletal muscle cell’s membrane potential; (d) the dynamics of the skeletal muscle cell’s attractor; (e) the temporal evolution of the Hamiltonian energy ratio between the skeletal muscle cell and the neuron; (f) the temporal change in the displacement of the active beam; (g) the temporal variation in the kinetic energy of the active beam; (h) the temporal evolution of the field energy within the memristor-coupled channel. In this analysis, the angular frequency is set to ω = 0.3. |
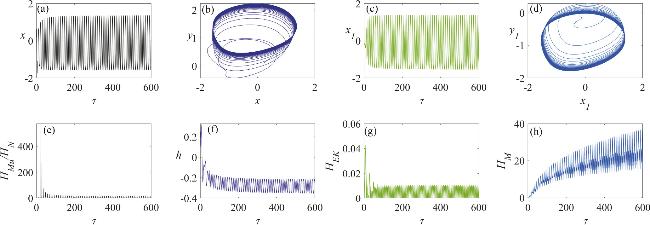
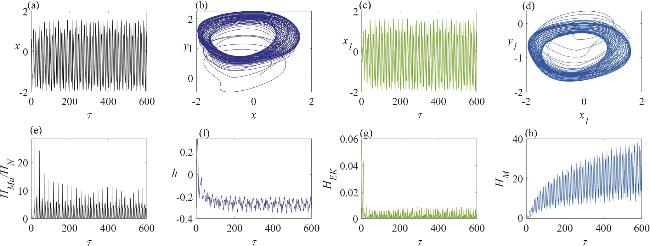
Figure 7. The dynamic characteristics of a neuron circuit and a skeletal muscle cell circuit in a chaotic firing state, under the influence of memristive synapse coupling: (a) the variation in the neuron’s membrane potential; (b) the dynamics of the neuron circuit’s attractor; (c) the variation in the skeletal muscle cell’s membrane potential; (d) the dynamics of the skeletal muscle cell’s attractor; (e) the temporal evolution of the Hamiltonian energy ratio between the skeletal muscle cell and the neuron; (f) the temporal change in the displacement of the active beam; (g) the temporal variation in the kinetic energy of the active beam; (h) the temporal evolution of the field energy within the memristor-coupled channel. In this analysis, the angular frequency is set to ω = 1.3. |
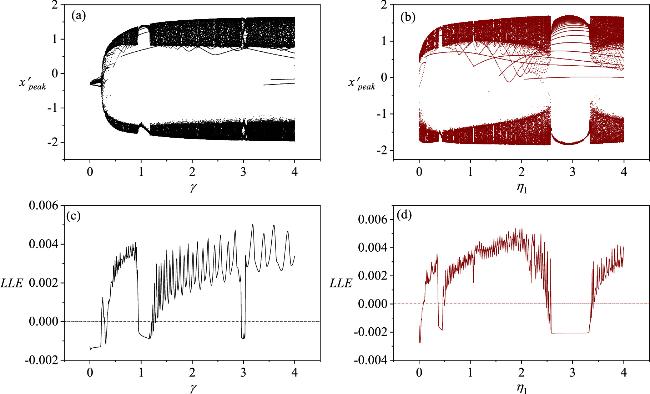
Figure 8. The bifurcation diagrams and maximum Lyapunov exponents for the variable direction parameters γ and η1 within the system where a neuron and a skeletal muscle cell are coupled via a memristor. In the diagrams, the parameters are assigned the following values: (a, c) η1 = 0.4;(b, d)γ = 1. The other parameters are fixed at a = 0.7, b = 0.3, c = 0.4, ξ = 0.175, I0 = 0.1, A = 0.4, λM = 0.1, α = 0.1, β = 0.1, η2 = 0.12, k1 = 0.1, k2 = 0.1, k3 = 0.1, m1 = 0.1, m2 = 0.1, m3 = 0.1, ω = 1.3. The system’s initial conditions are set to (0.2, 0.1, 0, 0.1, 0.1, 0.1, 0.1, 0.1). |
Figure 9. This figure illustrates the chaotic firing state of a memristor synapse-coupled system involving a neuron and a skeletal muscle cell: (a) the membrane potential of the neuron; (b) the attractor of the neuron circuit; (c) the membrane potential of the skeletal muscle cell; (d) the attractor of the skeletal muscle cell circuit; (e) the temporal evolution of the Hamiltonian energy ratio between the skeletal muscle cell and the neuron; (f) the temporal evolution of the displacement of the active beam; (g) the temporal evolution of the kinetic energy of the active beam; (h) the temporal evolution of the field energy in the memristor-coupled channel. The parameter values are set to γ = 3.2, η1 = 0.4. |
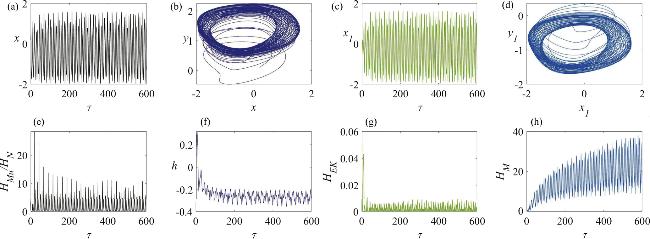
Figure 10. The chaotic firing state of a memristor synapse-coupled system involving a neuron and a skeletal muscle cell: (a) the membrane potential of the neuron; (b) the attractor of the neuron circuit; (c) the membrane potential of the skeletal muscle cell; (d) the attractor of the skeletal muscle cell circuit; (e) the temporal evolution of the Hamiltonian energy ratio between the skeletal muscle cell and the neuron; (f) the temporal evolution of the displacement of the active beam; (g) the temporal evolution of the kinetic energy of the active beam; (h) the temporal evolution of the field energy in the memristor-coupled channel. The parameter values are set to γ = 1.0, η1 = 2.0. |
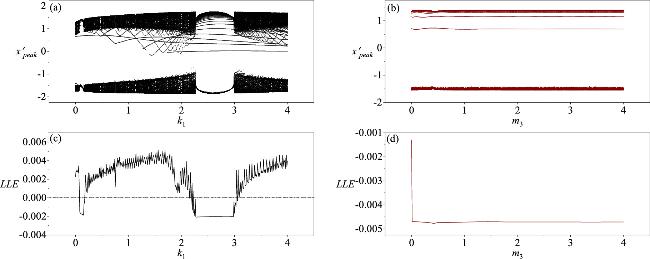
Figure 11. The bifurcation diagrams and Lyapunov exponents for the variable direction parameters k1 and m3 when a memristor synapse couples a neuron and a skeletal muscle cell. The parameters corresponding to the figures are: (a, c) m3 = 0.1; (b, d) k1 = 0.1; with a = 0.7, b = 0.3, c = 0.4, ξ = 0.175, I0 = 0.1, A = 0.4, λM = 0.1, α = 0.1, β = 0.1, γ = 3.2, η1 = 0.4, η2 = 0.12, k2 = 0.1, k3 = 0.1, m1 = 0.1, m2 = 0.1, ω = 1.3. The initial conditions of the system are (0.2, 0.1, 0, 0.1, 0.1, 0.1, 0.1, 0.1). |
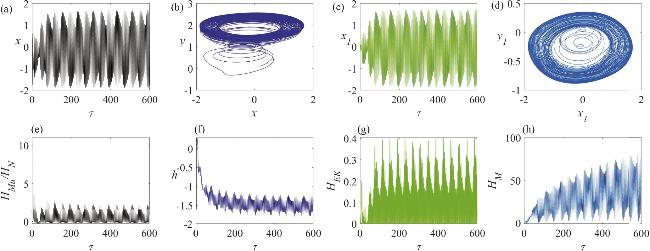
Figure 12. The chaotic firing state of a memristor synapse-coupled system involving a neuron and a skeletal muscle cell: (a) the membrane potential of the neuron; (b) the attractor of the neuron circuit; (c) the membrane potential of the skeletal muscle cell; (d) the attractor of the skeletal muscle cell circuit; (e) the temporal evolution of the Hamiltonian energy ratio between the skeletal muscle cell and the neuron; (f) the temporal evolution of the displacement of the active beam; (g) the temporal evolution of the kinetic energy of the active beam; (h) the temporal evolution of the field energy in the memristor-coupled channel. The parameter values are set to k1 = 1.8. |
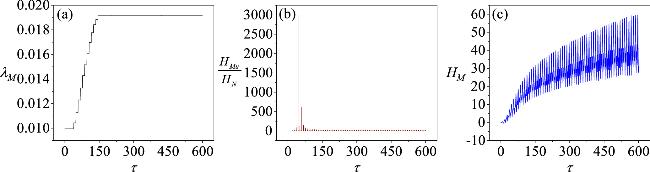
Figure 13. The evolution of coupling strength, the energy ratio between skeletal muscle cells and neurons, and the field energy in the coupling channel over time when the coupling strength is adaptively regulated. The parameters are selected as: a = 0.7, b = 0.3, c = 0.4, ξ = 0.175, I0 = 0.1, A = 0.4, α = 0.1, β = 0.1, γ = 1, η1 = 0.4, η2 = 0.12, k1 = 0.1, k2 = 0.1, k3 = 0.1, m1 = 0.1, m2 = 0.1, m3 = 0.1, m4 = 1, m5 = 0.1, σ = 0.1, ϵ = 25. The initial conditions are (0.2, 0.1, 0, 0.1, 0.1, 0.1, 0.1, 0.1). The initial value of the coupling strength λM(0) is 0.01. |