1. Introduction
2. Theory
2.1. Ab initio many-body perturbation theory
2.2. Other operators
2.2.1. Magnetic operator
2.2.2. β decay
2.2.3. Half-life
3. Results and discussion
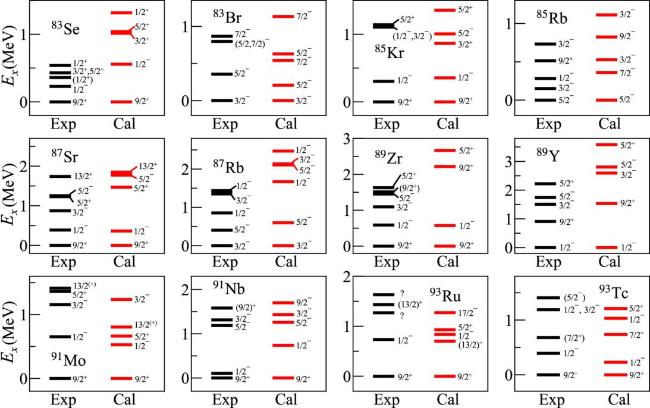
Figure 1. Spectra of N = 49 isotones and their daughter nuclei. Experimental data ara taken from [35]. The NN+3Nlnl interaction were used in the calculation. |
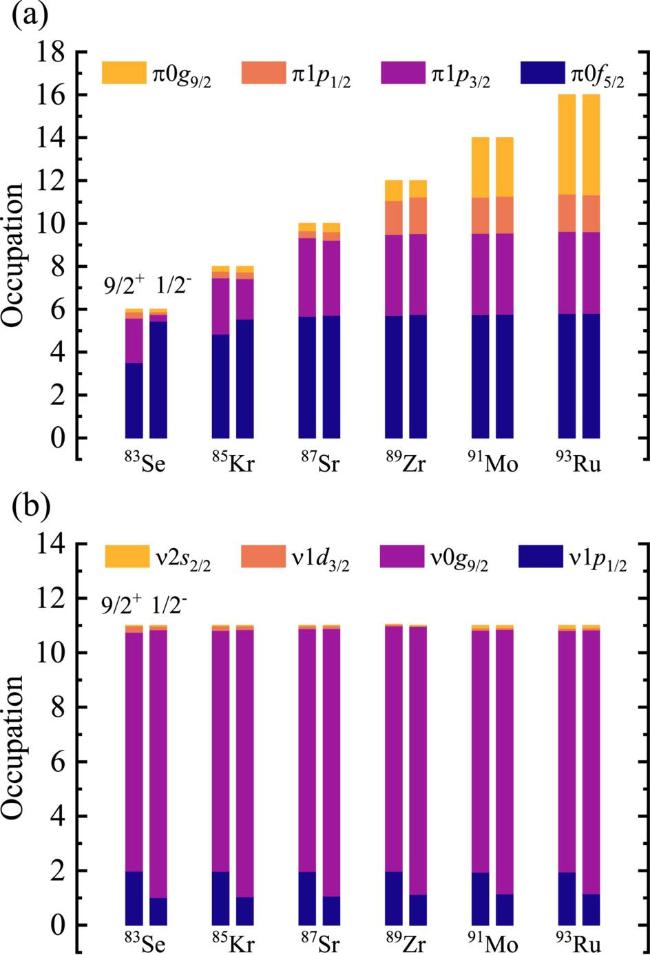
Figure 2. Configurations of 9/2+ states and 1/2− states in N = 49 isotones for proton orbits (a) and neutron orbits (b) in the valence-space. |
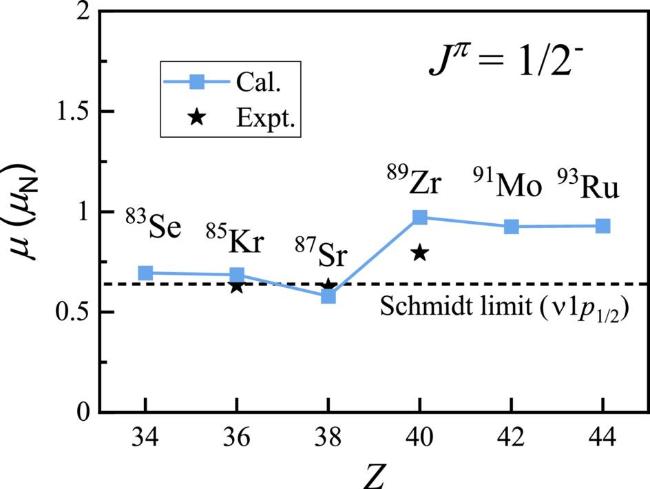
Figure 3. Nuclear magnetic moments for the 1/2− isomeric states of N = 49 isotones, compared with experimental data [35]. The horizontal dotted line means the single-particle value (Schmidt limit). |
Table 1. Calculated reduced transition probabilities B(M4) of the 1/2− isomers observed in N = 49 isotones in units of $1{0}^{5}{\mu }_{{\rm{N}}}^{2}{{\rm{fm}}}^{6}$. The NN+3Nlnl potentials are used. The experimental data is taken from [35]. |
| Nucleus | Transition | $B(M4)\,\left(1{0}^{5}{\mu }_{{\rm{N}}}^{2}{{\rm{fm}}}^{6}\right)$ | |
|---|---|---|---|
| Cal. | Expt. | ||
| 85Kr | ${\frac{1}{2}}^{-}\to {\frac{9}{2}}^{+}$ | 2.54 | 1.41(4) |
| 87Sr | 3.93 | 1.49(1) | |
| 89Zr | 4.41 | 1.58(1) | |
| 91Mo | 3.41 | 1.27(4) | |
| 93Ru | 2.77 | 1.18(14) | |
Table 2. Calculated β-decay reduced transition probabilities and calculated and experimental ${\mathrm{log}}\,ft$ values of the 1/2− isomers observed in 83Se, 85Kr and 87Sr. The NN+3Nlnl potentials are used. The experimental data is taken from [35]. |
| Transition | B(F) | B(GT) | ${\mathrm{log}}\,ft$ | |||
|---|---|---|---|---|---|---|
| Initial(Jπ) | Final(Jπ) | Expt. | Cal. | |||
| Expt. | Cal. | |||||
| 83Se(${\frac{1}{2}}_{1}^{-}$) | 83Br$\left({\frac{3}{2}}_{1}^{-}\right)$ | 83Br$\left({\frac{3}{2}}_{1}^{-}\right)$ | 0 | 7.29×10−4 | 6.0(1) | 6.9 |
| 83Br$\left({\frac{1}{2}}_{2}^{-},{\frac{3}{2}}_{3}^{-}\right)$ | 83Br$\left({\frac{1}{2}}_{2}^{-}\right)$ | 2.79×10−2 | 4.12×10−4 | 5.8(1) | 5.3 | |
| 83Br$\left({\frac{3}{2}}_{3}^{-}\right)$ | 0 | 1.42×10−2 | 5.6 | |||
| 83Br$\left({\left(\frac{3}{2}\right)}_{4}^{-}\right)$ | 83Br$\left({\frac{3}{2}}_{4}^{-}\right)$ | 0 | 2.30×10−3 | 5.4(1) | 6.4 | |
| 83Br$\left({\frac{1}{2}}_{3}^{-},{\frac{3}{2}}_{5}^{-}\right)$ | 83Br$\left({\frac{1}{2}}_{3}^{-}\right)$ | 2.31×10−4 | 5.76×10−4 | 7.3(1) | 6.9 | |
| 83Br$\left({\frac{3}{2}}_{5}^{-}\right)$ | 0 | 3.33×10−3 | 6.3 | |||
| 83Br$\left({\frac{1}{2}}_{4}^{-},{\frac{3}{2}}_{6}^{-}\right)$ | 83Br$\left({\frac{1}{2}}_{4}^{-}\right)$ | 2.72×10−5 | 4.25×10−7 | 6.0(1) | 8.4 | |
| 83Br$\left({\frac{3}{2}}_{6}^{-}\right)$ | 0 | 1.12×10−2 | 5.7 | |||
| 83Br$\left({\frac{1}{2}}_{5}^{(-)},{\frac{3}{2}}_{8}\right)$ | 83Br$\left({\frac{1}{2}}_{5}^{-}\right)$ | 2.41×10−4 | 5.99×10−4 | 5.9(1) | 6.9 | |
| 83Br$\left({\frac{3}{2}}_{8}^{-}\right)$ | 0 | 2.76×10−4 | 7.4 | |||
| 83Br$\left({\frac{1}{2}}_{6}^{-},{\frac{3}{2}}_{9}^{-}\right)$ | 83Br$\left({\frac{1}{2}}_{6}^{-}\right)$ | 1.36×10−3 | 2.51×10−3 | 4.8(1) | 6.2 | |
| 83Br$\left({\frac{3}{2}}_{9}^{-}\right)$ | 0 | 1.86×10−2 | 5.5 | |||
| 83Br$\left({\frac{1}{2}}_{10}^{-},{\frac{3}{2}}_{13}^{-}\right)$ | 83Br$\left({\frac{1}{2}}_{10}^{-}\right)$ | 8.28×10−3 | 8.75×10−3 | 5.9(4) | 5.6 | |
| 83Br$\left({\frac{3}{2}}_{13}^{-}\right)$ | 0 | 2.75×10−3 | 6.4 | |||
| 83Br$\left({\frac{1}{2}}_{11}^{-},{\frac{3}{2}}_{14}^{-}\right)$ | 83Br$\left({\frac{1}{2}}_{11}^{-}\right)$ | 3.60×10−2 | 4.40×10−2 | 5.7(1) | 4.9 | |
| 83Br$\left({\frac{3}{2}}_{14}^{-}\right)$ | 0 | 8.53×10−2 | 4.9 | |||
| 85Kr$\left({\frac{1}{2}}_{1}^{-}\right)$ | 85Rb$\left({\frac{3}{2}}_{1}^{-}\right)$ | 85Rb$\left({\frac{3}{2}}_{1}^{-}\right)$ | 0 | 5.71×10−2 | 7.1(1) | 5.042 |
| 85Rb$\left({\frac{1}{2}}_{1}^{-}\right)$ | 85Rb$\left({\frac{1}{2}}_{1}^{-}\right)$ | 4.17×10−5 | 6.51×10−4 | 7.39(2) | 6.96 | |
| 85Rb$\left({\frac{3}{2}}_{2}^{-}\right)$ | 85Rb$\left({\frac{3}{2}}_{2}^{-}\right)$ | 0 | 2.98×10−5 | 5.250(8) | 8.325 | |
| 87Sr$\left({\frac{1}{2}}_{1}^{-}\right)$ | 87Rb$\left({\frac{3}{2}}_{1}^{-}\right)$ | 87Rb$\left({\frac{3}{2}}_{1}^{-}\right)$ | 0 | 1.51 | 4.40(12) | 3.62 |
Table 3. Same as table 2 but for 1/2− isomers in 89Zr, 91Mo and 93Ru. |
| Transition | B(F) | B(GT) | ${\mathrm{log}}\,ft$ | |||
|---|---|---|---|---|---|---|
| Initial(Jπ) | Final(Jπ) | Expt. | Cal. | |||
| Expt. | Cal. | |||||
| 89Zr$\left({\frac{1}{2}}_{1}^{-}\right)$ | 89Y$\left({\frac{1}{2}}_{1}^{-}\right)$ | 89Y$\left({\frac{1}{2}}_{1}^{-}\right)$ | 1.23×10−2 | 3.36×10−2 | 7.1(4) | 5.1 |
| 89Y$\left({\frac{3}{2}}_{1}^{-}\right)$ | 89Y$\left({\frac{3}{2}}_{1}^{-}\right)$ | 0 | 1.63 | 4.31(2) | 3.59 | |
| 91Mo$\left({\frac{1}{2}}_{1}^{-}\right)$ | 91Nb$\left({\frac{1}{2}}_{1}^{-}\right)$ | 91Nb$\left({\frac{1}{2}}_{1}^{-}\right)$ | 7.56×10−3 | 2.88×10−2 | 5.94(22) | 1.27(4) |
| 91Nb$\left({\frac{3}{2}}_{1}^{-}\right)$ | 91Nb$\left({\frac{3}{2}}_{1}^{-}\right)$ | 0 | 3.31×10−3 | 4.78(4) | ||
| 91Nb$\left({\frac{3}{2}}_{2}^{-}\right)$ | 91Nb$\left({\frac{3}{2}}_{2}^{-}\right)$ | 0 | 1.54 | 4.4(4) | ||
| 91Nb$\left({\left(\frac{3}{2}\right)}_{3}^{-}\right)$ | 91Nb$\left({\frac{3}{2}}_{3}^{-}\right)$ | 0 | 5.11×10−3 | 5.03(4) | ||
| 93Ru$\left({\frac{1}{2}}_{1}^{-}\right)$ | 93Tc$\left({\frac{1}{2}}_{1}^{-}\right)$ | 93Tc$\left({\frac{1}{2}}_{1}^{-}\right)$ | 1.27×10−2 | 3.93×10−2 | >5.6 | 5.1 |
| 93Tc$\left({\frac{1}{2}}_{3}^{-},{\frac{3}{2}}_{2}^{-}\right)$ | 93Tc$\left({\frac{1}{2}}_{3}^{-}\right)$ | 1.20×10−2 | 1.05×10−2 | 4.84(3) | 5.45 | |
| 93Tc$\left({\frac{3}{2}}_{2}^{-}\right)$ | 0 | 2.28×10−3 | 6.44 | |||
| 93Tc$\left({\frac{1}{2}}_{4}^{-},{\frac{3}{2}}_{3}^{-}\right)$ | 93Tc$\left({\frac{1}{2}}_{4}^{-}\right)$ | 9.29×10−3 | 2.93×10−3 | 4.526(19) | 5.712 | |
| 93Tc$\left({\frac{3}{2}}_{3}^{-}\right)$ | 0 | 1.38 | 3.658 | |||
| 93Tc$\left({\left({\frac{1}{2}}_{5},{\frac{3}{2}}_{4}\right)}^{-}\right)$ | 93Tc$\left({\frac{1}{2}}_{4}^{-}\right)$ | 1.75×10−2 | 3.51×10−3 | 4.72(3) | 5.48 | |
| 93Tc$\left({\frac{3}{2}}_{4}^{-}\right)$ | 0 | 3.16×10−2 | 5.30 | |||
| 93Tc$\left({\left(\frac{3}{2}\right)}_{5}^{-}\right)$ | 93Tc$\left({\frac{3}{2}}_{5}^{-}\right)$ | 0 | 2.06×10−3 | ≤4.7 | 6.5 | |
Table 4. Calculated half-life of the 1/2− isomers in N = 49 isotones, compared with experimental [35]. |
| Nucleus | half-life | |
|---|---|---|
| Cal. | Expt. | |
| 83Se | 2.74 min | 1.17(1) min |
| 85Kr | 6.197 h | 4.480(8) h |
| 87Sr | 3.645 h | 2.815(12) h |
| 89Zr | 32.638 min | 4.161(12) min |
| 91Mo | 76.1 s | 65.0(7) s |
| 93Ru | 7.4 s | 10.8(3) s |