1. Introduction
2. Model
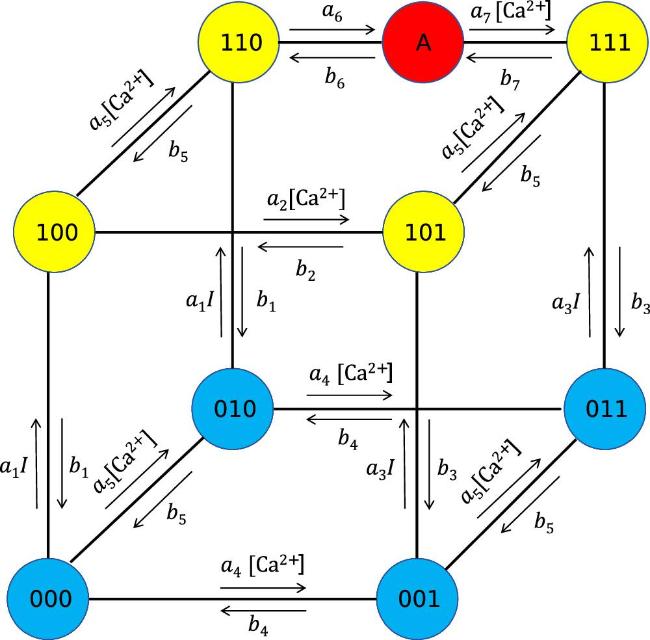
Figure 1. Nine-state model of the IP3R channel classified by binding status. Blue-filled circles represent states where IP3 is unbound, yellow-filled circles indicate states where IP3 is bound, and red-filled circles denote the fully activated state A. The 110 state is the pre-activated state. Arrows represent state transitions, with transition rates influenced by IP3 and Ca2+ concentrations. |
Table 1. Parameter values used in the nine-state model of IP3R. Parameters highlighted in bold are newly introduced in our model and are fitted based on data from [27]. All other parameters are adopted from the DYK model [17]. The symbol δr denotes the change in the Pearson correlation coefficient resulting from a 50% variation of the corresponding parameter. |
| Parameter | Value | Unit | δr | Description |
|---|---|---|---|---|
| a1 | 400 | μM−1s−1 | 0.006 | IP3 (binding) |
| a2 | 0.2 | μM−1s−1 | 0.002 | Ca2+ (binding) |
| a3 | 400 | μM−1s−1 | 0.030 | IP3 (binding) |
| a4 | 0.2 | μM−1s−1 | 0.001 | Ca2+ (binding) |
| a5 | 20 | μM−1s−1 | 0.094 | Ca2+ (binding) |
| a6 | 80 | s−1 | 0.111 | Ca2+ (activation) |
| a7 | 0.2 | μM−1s−1 | 0.089 | Ca2+ (inhibition) |
| b1 | 52 | s−1 | 0.004 | IP3 (unbinding) |
| b2 | 0.2098 | s−1 | 0.031 | Ca2+ (unbinding) |
| b3 | 377.36 | s−1 | 0.019 | IP3 (unbinding) |
| b4 | 0.0289 | s−1 | 0.006 | Ca2+ (unbinding) |
| b5 | 1.6468 | s−1 | 0.070 | Ca2+ (unbinding) |
| b6 | 100 | s−1 | 0.093 | Ca2+ (inactivation) |
| b7 | 0.2098 | s−1 | 0.040 | Ca2+ (activation) |
Table 2. Parameters for the exchange of calcium between the cytosol and the ER [17]. |
| Parameter | Value | Description |
|---|---|---|
| c0 | 2.0 μM | Total [Ca2+] in terms of cytosolic volume |
| c1 | 0.185 | (ER volume)/(cytosolic volume) |
| ν1 | 6 s−1 | Max Ca2+ channel flux |
| ν2 | 0.11 s−1 | Ca2+ leak flux constant |
| ν3 | 0.9 μM−1s−1 | Max Ca2+ uptake |
| k3 | 0.1 μM | Activation constant for ATP-Ca2+ pump |
3. Results
3.1. Methodology
3.2. Distribution of IP3R states under saturated IP3 conditions
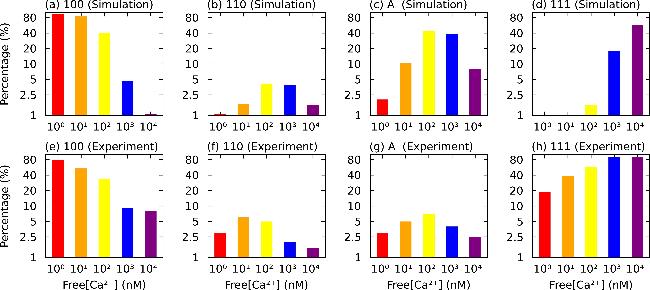
Figure 2. State distribution at different fixed Ca2+ concentrations under saturated IP3 condition. The patterns in a, b, c, and d depict the simulated state distributions of the 100, 110, A, and 111 states at varying orders of magnitude of Ca2+ concentration, respectively. The patterns in e, f, g, and h show the corresponding state distributions reported from the results of EM experiments [27]. Both the simulations and experiments were conducted with IP3 = 2 μM. |
3.3. Open probability at different IP3R concentrations and role of IP3 state
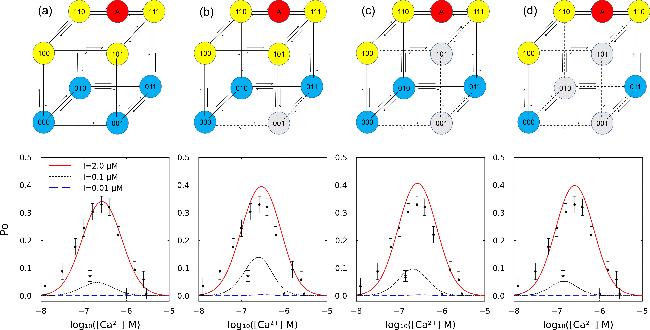
Figure 3. Comparison of the nine-state model and three simplified models obtained by sequentially removing the 001, 101, and 010 states (gray indicates removed states). The lower panel shows the simulated open probability (PO) for each model alongside experimental data from single-channel patch-clamp recordings of IP3R on the native nuclear membrane. Circles represent data for [IP3] = 0.2 μM, while stars indicate data for [IP3] = 0.01 μM [39, 40]. |
3.4. Calcium oscillation
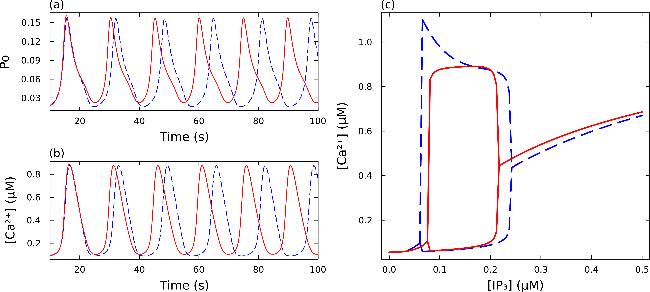
Figure 4. Calcium oscillations. (a) Variation in open probability over time. (b) Variation in calcium concentration [Ca2+] over time. (c) Bifurcation diagram of calcium concentration as a function of [IP3] levels. In all panels, the red solid line represents the nine-state model and the blue dashed line represents the six-state model. |