1. Introduction
2. Gradient-conservative structure
2.1. Basic formulation
2.2. Generalized gradient systems
2.3. Relation to the large deviations principle
2.4. Interpretation of gradient flows in Wasserstein space
Given two functions ${h}_{1},{h}_{2}:{\rm{\Omega }}\to {\mathbb{R}}$ with $\int$Ωh1dx = $\int$Ωh2dx = 0, one can define their Wasserstein scalar product at ρ as
Given a functional $F:{{ \mathcal P }}_{2}({\rm{\Omega }})\to {\mathbb{R}}$, its gradient with respect to the Wasserstein scalar product at $\bar{\rho }\in {{ \mathcal P }}_{2}({\rm{\Omega }})$ is the unique function ${{\rm{\nabla }}}_{{W}_{2}}F(\bar{\rho })$ (if it exists) such that
Given a functional $F:{{ \mathcal P }}_{2}({\rm{\Omega }})\to {\mathbb{R}}$, a curve of probability measure $\rho :[0,T)\to {{ \mathcal P }}_{2}({\rm{\Omega }})$ is a gradient flow of F with respect to W2 and with starting point ${\bar{\rho }}_{0}$ if
3. Unifying various thermodynamically compatible modeling approaches
3.1. GENERIC
3.2. Onsager's variational principle
3.3. Energetic variational approach
3.4. Classical irreversible thermodynamics and beyond
Table 1. Summary of thermodynamically compatible modeling approaches. |
| Approach | Key elements | Governing equations |
|---|---|---|
| Conservative dynamics | Energy function E(z), antisymmetric matrix A(z) | $\frac{{\rm{d}}{\boldsymbol{z}}}{{\rm{d}}t}={\boldsymbol{A}}({\boldsymbol{z}})\cdot {\rm{\nabla }}E({\boldsymbol{z}})$ |
| Hamiltonian dynamics | Hamiltonian function H(z, ξ) | $\frac{{\rm{d}}{\boldsymbol{z}}}{{\rm{d}}t}=\frac{\partial H}{\partial {\boldsymbol{\xi }}},\frac{{\rm{d}}{\boldsymbol{\xi }}}{{\rm{d}}t}=-\frac{\partial H}{\partial {\boldsymbol{z}}}$ |
| Lagrangian dynamics | Lagrangian function $L({\boldsymbol{z}},\dot{{\boldsymbol{z}}})$ | $\frac{{\rm{d}}}{{\rm{d}}t}\left(\frac{\partial L}{\partial \dot{{\boldsymbol{z}}}}\right)-\frac{\partial L}{\partial {\boldsymbol{z}}}={\bf{0}}$ |
| | ||
| Gradient flow | Free energy F(z) | $\frac{{\rm{d}}{\boldsymbol{z}}}{{\rm{d}}t}=-{\rm{\nabla }}F({\boldsymbol{z}})$ |
| Dissipative dynamics | Free energy F(z), dissipation matrix M(z) | $\frac{{\rm{d}}{\boldsymbol{z}}}{{\rm{d}}t}=-{\boldsymbol{M}}({\boldsymbol{z}})\cdot {\rm{\nabla }}F({\boldsymbol{z}})$ |
| Generalized gradient systems | Free energy F(z), dissipative functions $\psi ({\boldsymbol{z}},\dot{{\boldsymbol{z}}}),{\psi }^{* }({\boldsymbol{z}},{\boldsymbol{\xi }})$ | $\begin{array}{l}\psi ({\boldsymbol{z}},\dot{{\boldsymbol{z}}})+{\psi }^{* }({\boldsymbol{z}},-{\rm{\nabla }}F)\\ +\langle {\rm{\nabla }}F,\dot{{\boldsymbol{z}}}\rangle =0\end{array}$ |
| Onsager variational principle | Free energy F(z), dissipative function $\psi ({\boldsymbol{z}},\dot{{\boldsymbol{z}}})$ | $\begin{array}{l}\frac{\partial { \mathcal R }({\boldsymbol{z}},\dot{{\boldsymbol{z}}})}{\partial \dot{{\boldsymbol{z}}}}={\bf{0}},\\ { \mathcal R }({\boldsymbol{z}},\dot{{\boldsymbol{z}}})=\psi ({\boldsymbol{z}},\dot{{\boldsymbol{z}}})+\langle {\rm{\nabla }}F,\dot{{\boldsymbol{z}}}\rangle \end{array}$ |
| Energetic variational approach | Action ${A}_{F}({\boldsymbol{z}},\dot{{\boldsymbol{z}}})$, dissipative function $\psi ({\boldsymbol{z}},\dot{{\boldsymbol{z}}})$ | $\begin{array}{l}\frac{\partial {A}_{F}({\boldsymbol{z}},\dot{{\boldsymbol{z}}})}{\partial {\boldsymbol{z}}}=\frac{\partial \psi ({\boldsymbol{z}},\dot{{\boldsymbol{z}}})}{\partial \dot{{\boldsymbol{z}}}},\\ {A}_{F}={\displaystyle \int }_{0}^{t}(K-F){\rm{d}}\tau \end{array}$ |
4. Applications
4.1. Chemical mass-action equations
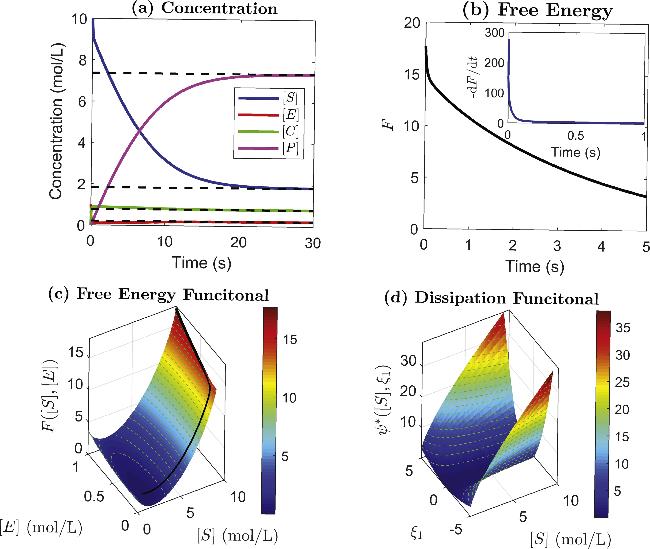
Figure 1. Dynamic and thermodynamic structures of the MM reactions. (a) The concentrations versus time of the species S, E, C, and P; the solid lines represent temporal states, while the dashed lines represent equilibrium states. (b) The free energy function F(t) and its negative gradient fd(t) (inset) versus time. (c) The free energy surface F([S], [E]) versus the concentrations [S] and [E], while the black solid line represents the trajectory from the given initial state. (d) The dissipation function ψ*([S], ξ1) versus the concentration [S] and ξ1. For all plots, the initial concentrations are ([S], [E], [C], [P])|t=0 = (10, 1, 0, 0), and the rate constants are $({\kappa }_{1}^{+},{\kappa }_{1}^{-},{\kappa }_{2}^{+},{\kappa }_{2}^{-})=(2,1,1,0.5)$. |
4.2. Diffusion equations
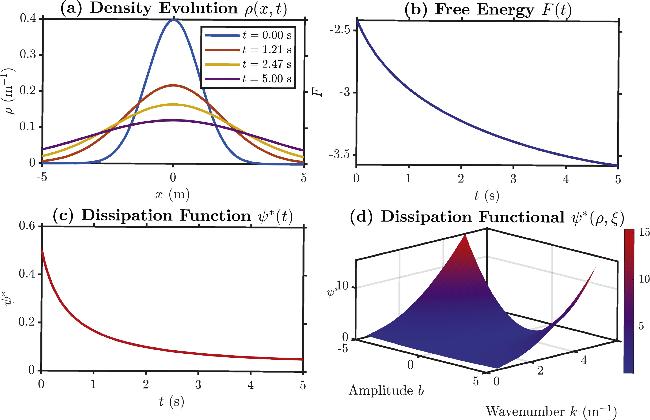
Figure 2. Dynamic and thermodynamic structures of the diffusion equations. (a) Density profiles ρ(t, x) at four different times. (b) Temporal evolution of the free energy function $F(t)=\int (\rho {\rm{ln}}\rho -\rho )dx$. (c) Temporal evolution of the dissipation function ${\psi }^{* }(t)=\int \frac{1}{2}D\rho | {\rm{\nabla }}\xi {| }^{2}dx$ along the path $\xi =-{{\rm{\nabla }}}_{\rho }F=-{\rm{ln}}\rho $. (d) Dissipation functional surface ψ*(ρ, ξ) at fixed t = 2.47 with $\xi (x)=b\cdot {\rm{\sin }}(2\pi kx/L)$, illustrating its dependence on the amplitude b and wavenumber k. For all plots, the diffusion coefficient D = 1.0, spatial domain [-L, L] with L = 20. |